Traceability Overview
What is Traceability?
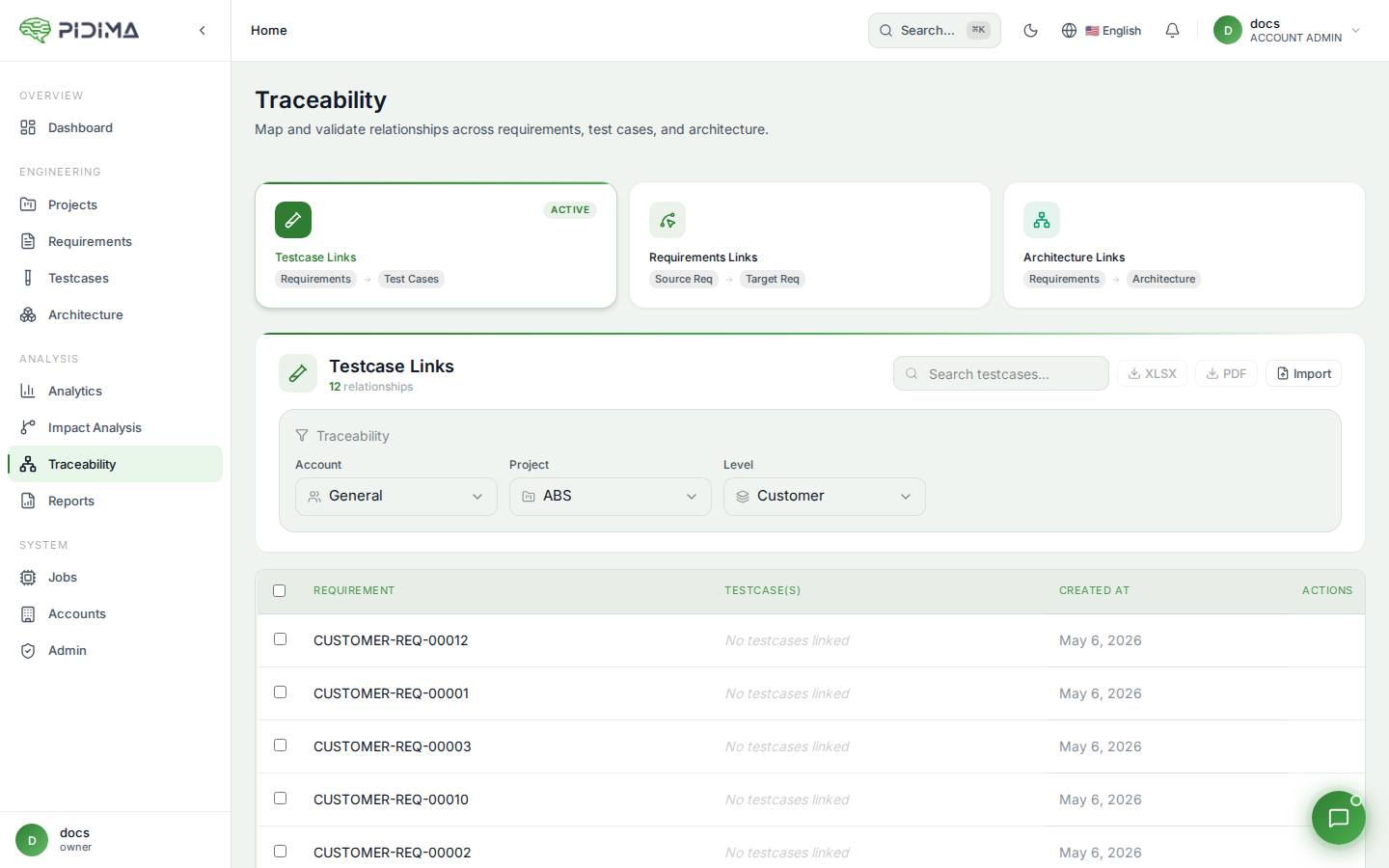
Traceability in Pidima creates documented relationships between:
- Requirements and Testcases: Track which testcases validate each requirement
- Bidirectional Links: Navigate from requirements to testcases and vice versa
- Coverage Documentation: Prove all requirements have adequate test coverage
Why Traceability Matters
Regulatory Compliance:
- FDA requires traceability for medical devices
- ISO standards mandate documented test coverage
- Aerospace and automotive industries require full traceability
Project Benefits:
- Ensure complete test coverage
- Identify untested requirements
- Demonstrate compliance readiness
- Support audit documentation